
Waleed Javaid, an associate professor of infectious disease at Icahn School of Medicine at Mount Sinai in New York City, he said. So you have more than likely wasted the first dose, if you don’t get the second one in time,” Dr. “Your immunity is incomplete, and it might not be effective. If you experience mild to moderate side effects after the first dose, that doesn’t mean you should skip the second dose. It’s important to recognize that some side effects from the Pfizer-BioNTech COVID-19 vaccine are common and normal. We need to continue to collect data to make sure it stays that way,” he said. 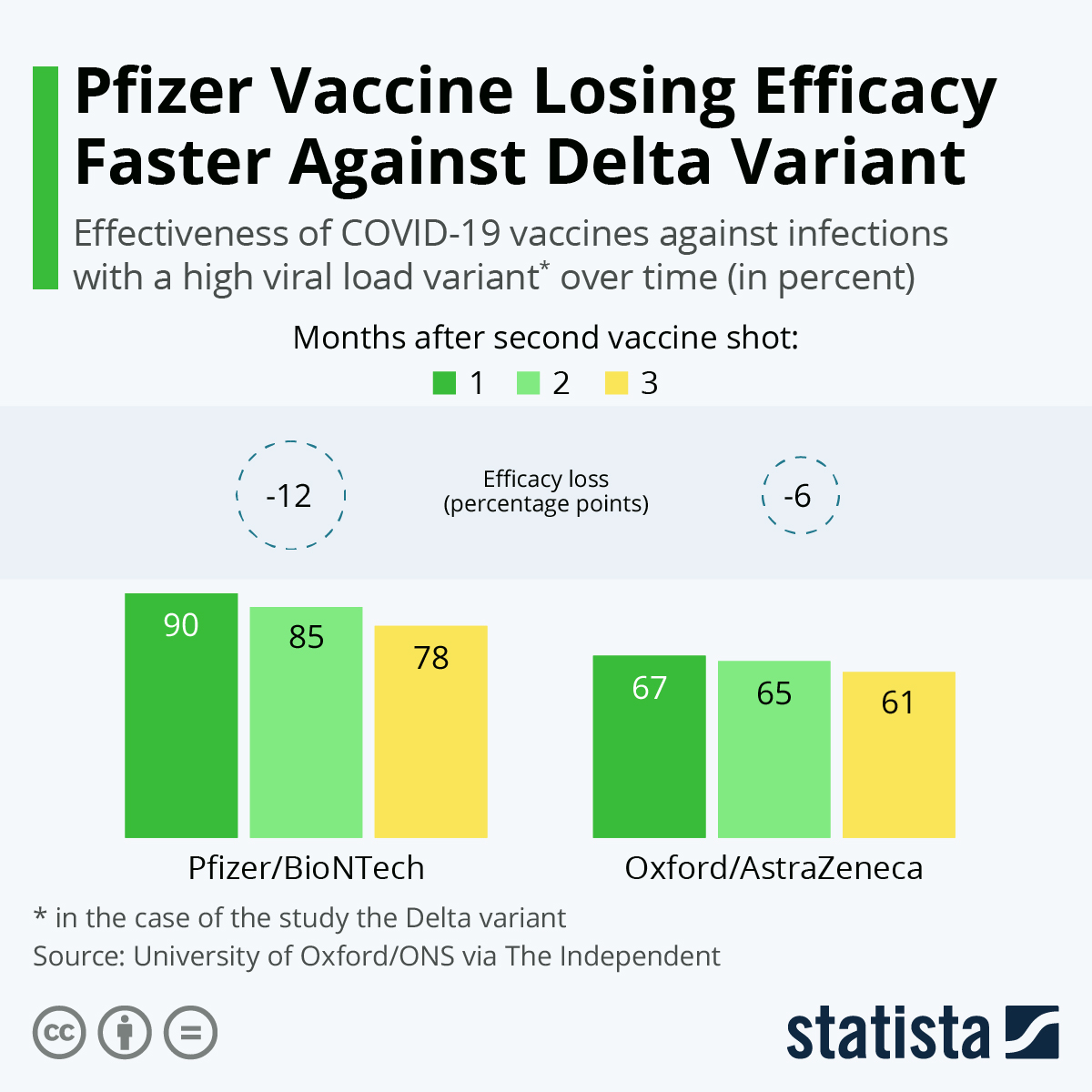
“Of course, we have to continue to monitor going forward. Matthew Heinz, a hospitalist based in Tucson, Arizona, told Healthline. “Based on the large number of folks that have been closely monitored during the study, the patient safety profile is excellent in terms of side effects,” Dr. So far, research has also found that the vaccine has a good safety profile. The available data suggests that after two doses, the vaccine is 95 percent effective at preventing COVID-19. Based on evidence from ongoing clinical trials, the FDA found that the known and potential benefits of the vaccine outweigh the known and potential risks. The FDA issued this authorization after reviewing the available efficacy and safety data on the new vaccine. Under this EUA, Pfizer-BioNTech’s new COVID-19 vaccine can now be distributed in the United States. Use of COVID-19 Vaccines in the U.S.Earlier this month, the Food and Drug Administration issued its first emergency use authorization (EUA) for a vaccine against COVID-19 in people aged 16 years and older.Interim Clinical Considerations plus icon.The COVID-19 vaccination schedules for People who are not moderately or severely immunocompromised and People who are moderately or severely immunocompromised should be consulted for age-specific information see also Appendix C for recommended actions following interchangeability-related errors or deviations in administration of COVID-19 vaccines. Ages 6 years and older: People who are unvaccinated or previously received 1 or more doses of any monovalent COVID-19 vaccine are authorized to receive either bivalent Moderna or bivalent Pfizer-BioNTech COVID-19 vaccine.įor additional information, see Interchangeability of COVID-19 vaccines.Monovalent Pfizer-BioNTech COVID-19 are authorized to receive only bivalent Pfizer-BioNTech COVID-19 Vaccine.Monovalent Moderna COVID-19 Vaccine are authorized to receive either bivalent Moderna or bivalent Pfizer-BioNTech COVID-19 vaccine. 
Age 5 years: Children who are unvaccinated or previously received 1 or more doses of:.Ages 6 months–4 years: Children who are unvaccinated or previously received 1 or more doses of a monovalent mRNA vaccine are authorized to receive only bivalent mRNA vaccine dose(s) from the same vaccine manufacturer.Use of mRNA COVID-19 vaccines interchangeably from different manufacturers (Moderna and Pfizer-BioNTech) varies by recipient age, vaccination history, and vaccine product: Coadministration of COVID-19 vaccines with other vaccines.Interim Clinical Considerations for Use of JYNNEOS and ACAM2000 Vaccines during the 2022 U.S.However, if a patient’s risk for mpox or severe disease due to COVID-19 is increased, administration of mpox and COVID-19 vaccines should not be delayed. This is because of the observed risk for myocarditis and pericarditis after receipt of ACAM2000 orthopoxvirus vaccine and COVID-19 vaccines, and the hypothetical risk for myocarditis and pericarditis after JYNNEOS vaccine. People, particularly adolescent or young adult males, who are recommended to receive both vaccines might consider waiting 4 weeks between vaccines.Use of JYNNEOS vaccine should be prioritized over ACAM2000 when co-administering a COVID-19 vaccine and an orthopoxvirus vaccine.There is no required minimum interval between receiving a dose of any COVID-19 vaccine and an orthopoxvirus vaccine, either JYNNEOS or ACAM2000 vaccine (e.g., for mpox prevention), regardless of which vaccine is administered first.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
